THROMBATE III provides a direct approach to managing hATd in high-risk situations1
Clinical studies have shown that THROMBATE III is an effective choice for adult and pediatric patients with hATd for the treatment and prevention of thromboembolism, including before, during, and after surgery and childbirth.1
Trusted for more than 30 years1
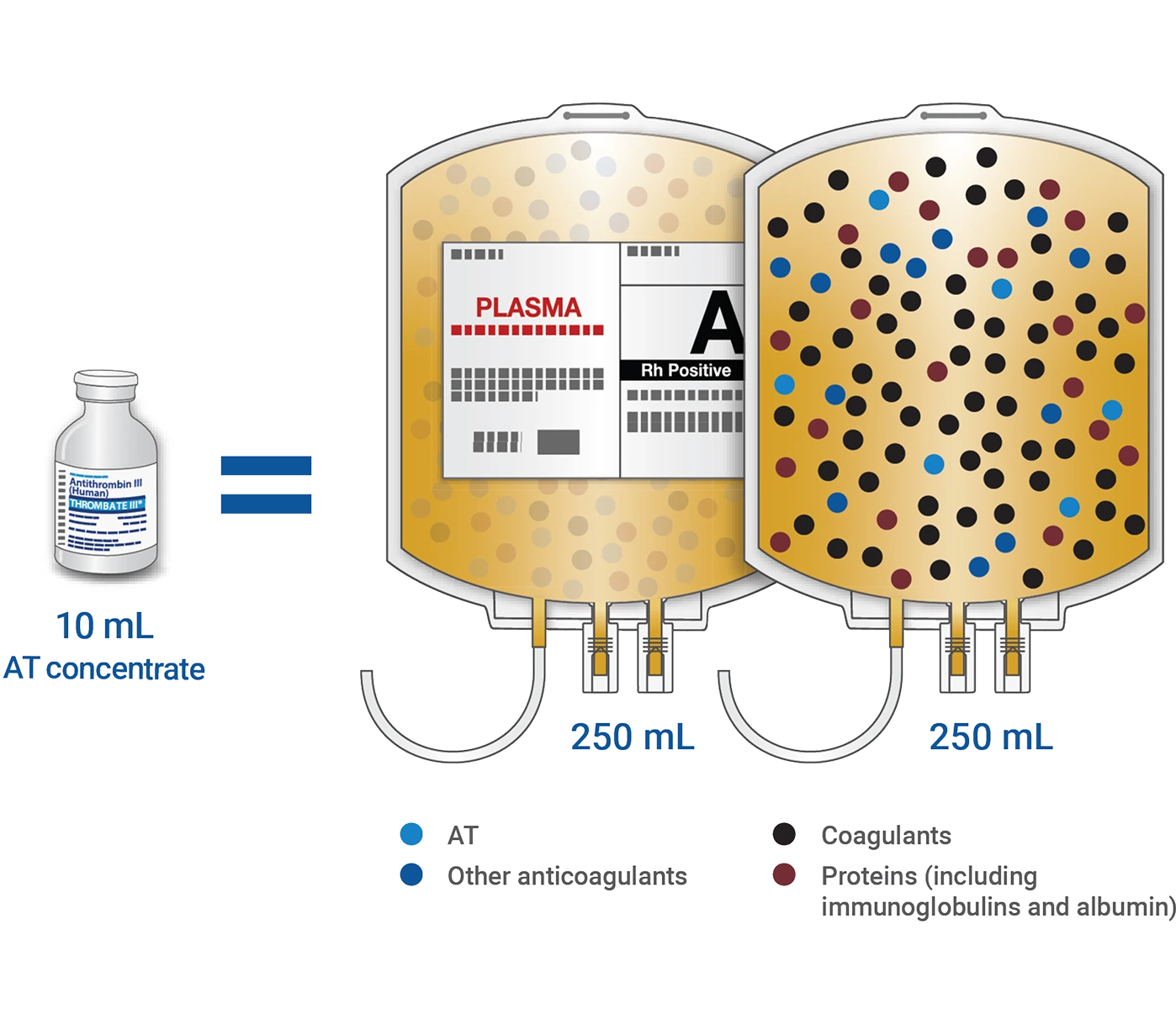
THROMBATE III delivers 50x more antithrombin (AT) than the same amount of fresh frozen plasma (FFP)—and none of the additional proteins and factors1,2
Lower volume
Just one 10-mL vial of THROMBATE III has the same IU of AT as 500 mL of FFP3
Cost-effective
THROMBATE III may be a cost-effective and timely option compared to FFP when considering administration, preparation, and additional expenses3,*
*Additional expenses associated with FFP may include overhead, transportation, and defrosting time.
Properties of AT replacement and FFP
- THROMBATE III1
Indicated in adult and pediatric patients with hATd for treatment and prevention of thromboembolism and for prevention of perioperative and peripartum thromboembolism
- FFP13
Indicated in the management of patients with selected coagulation factor deficiencies, congenital or acquired, for which no specific coagulation concentrates are available
- THROMBATE III1
Single-use 10-mL vial (500-IU potency)
- FFP13
Supplied in 200- to 250-mL bags (on average)
- THROMBATE III1
50 IU/mL AT concentration (after reconstitution with 10 mL sterile water for injection)
- FFP13
~1 IU/mL AT concentration
- THROMBATE III1
Intravenous bolus infusion, regardless of ABO status. Can be readily available at the point of care, no thawing required
- FFP13
Needs to be thawed prior to use. Intravenous infusion. Plasma must be ABO-compatible with the recipient's red blood cells
- THROMBATE III1
THROMBATE III Provides predictable amounts of AT
- FFP13
Contains AT plus other plasma components in varying levels
- THROMBATE III1
The half-life of THROMBATE III is similar to endogenous AT1,4
- FFP13
The components of FFP have varying half-lives5
- THROMBATE III1
THROMBATE III can be stored at room temperature (up to 77°F) for up to 36 months. Do not freeze
- FFP13
FFP should be stored at −18°C (0°F) or colder. Infuse immediately after thawing or store at 1-6°C (34-43°F)
- THROMBATE III1
THROMBATE III is produced from human plasma— it is fractionated and purified to yield concentrated antithrombin
- FFP13
Centrifuged, separated, and frozen solid at −18°C (0°F) within 8 hours of collection
- THROMBATE III1
The loading dose for THROMBATE III is calculated with a clear formula:
Units required (IU) = 120% - baseline % x body weight (kg)/1.4%
- FFP13
The volume of FFP transfused depends on various factors, including the clinical situation and patient weight, and may be guided by laboratory assays of coagulation function
| THROMBATE III1 | FFP13 | |
|---|---|---|
|
Indication |
Indicated in adult and pediatric patients with hATd for treatment and prevention of thromboembolism and for prevention of perioperative and peripartum thromboembolism |
Indicated in the management of patients with selected coagulation factor deficiencies, congenital or acquired, for which no specific coagulation concentrates are available |
|
How supplied/volume |
Single-use 10-mL vial (500-IU potency) |
Supplied in 200- to 250-mL bags (on average) |
|
Concentration |
50 IU/mL AT concentration (after reconstitution with 10 mL sterile water for injection) |
~1 IU/mL AT concentration |
|
Use |
Intravenous bolus infusion, regardless of ABO status. Can be readily available at the point of care, no thawing required |
Needs to be thawed prior to use. Intravenous infusion. Plasma must be ABO-compatible with the recipient's red blood cells |
|
Content |
THROMBATE III Provides predictable amounts of AT |
Contains AT plus other plasma components in varying levels |
|
Half-life |
The half-life of THROMBATE III is similar to endogenous AT1,4 |
The components of FFP have varying half-lives5 |
|
Storage |
THROMBATE III can be stored at room temperature (up to 77°F) for up to 36 months. Do not freeze |
FFP should be stored at −18°C (0°F) or colder. Infuse immediately after thawing or store at 1-6°C (34-43°F) |
|
Process |
THROMBATE III is produced from human plasma— it is fractionated and purified to yield concentrated antithrombin |
Centrifuged, separated, and frozen solid at −18°C (0°F) within 8 hours of collection |
|
Dosing |
The loading dose for THROMBATE III is calculated with a clear formula: |
The volume of FFP transfused depends on various factors, including the clinical situation and patient weight, and may be guided by laboratory assays of coagulation function |
The half-life of AT has been reported to be shortened following surgery, hemorrhage, or acute thrombosis, and during intravenous heparin (or low molecular weight heparin) administration. In such conditions, monitor plasma AT levels more frequently, and administer THROMBATE III as necessary.
THROMBATE III is effective in preventing the expansion of a formed thrombus (clot) and formation of additional clots in patients with hATd1
No reports of thrombotic complications during obstetrical and surgical procedures¹
- # of Procedures*
N = 13
- Outcome
No thrombosis or pulmonary embolism*
| Clinical Situation | # of Procedures* | Outcome |
|---|---|---|
|
Childbirth/Surgery |
N = 13 |
No thrombosis or pulmonary embolism* |
Patients diagnosed with hATd are at especially high risk for blood clots in certain situations such as surgery, pregnancy, and childbirth, and when they already have a blood clot.6
Perform coagulation tests to avoid excessive or insufficient anticoagulation and monitor for bleeding or thrombosis. Measure functional plasma AT levels with amidolytic or clotting assays, do not use immunoassays.
In clinical studies, the most common adverse reactions (≥5% of patients) were dizziness, chest discomfort, nausea, dysgeusia, and pain (cramps).
According to the American Society of Hematology (ASH) for the management of pediatric VTE:
AT replacement therapy is recommended in children with documented hATd failing to respond to anticoagulation therapy for VTE, as well as use in addition to standard anticoagulation in pediatric patients with deep vein thrombosis, cerebral sinovenous thrombosis, and pulmonary embolism who have clinically failed to achieve therapeutic anticoagulation with persistent low AT levels.7

THROMBATE III offers safe, convenient, and predictable dosing1
Calculating the right dosage* is easy
- Has one dosing formula1
- Bolus intravenous infusion‡
- Loading dose on day 1
Dosing calculator
Calculate the loading dose for Thrombate III.
Loading dose
Please enter your patient's information to calculate the appropriate dose.
THROMBATE III is available in single-use 10-mL vials (500-IU potency) through all major distributors
NDC numbers to order:
- 13533-602-50
- 13533-606-12
Provides for convenient storage and reconstitution1
- No refrigeration required
- Store at room temperature (not to exceed 25 °C, 77 °F)
- No need to thaw
- Sterile water for injection (10 mL), transfer needle, and filter needle provided
Hypersensitivity reactions may occur. Should evidence of an acute hypersensitivity reaction be observed, promptly interrupt the infusion and begin appropriate treatment.
*For complete details, review the dosing table provided in the full Prescribing Information for THROMBATE III.
†Expressed as a percent of normal level based on functional AT assay. If laboratory testing is available, monitor plasma AT levels every 12 hours following the initial loading dose. THROMBATE III may be infused intravenously over 10 to 20 minutes.
‡Rate of administration adapted to the response of the individual patient, but administration of the entire dose in 10 to 20 minutes is generally well tolerated.
The risks of low AT
See how THROMBATE III replaces missing AT
Learn more about THROMBATE III
Have a question about THROMBATE III?
Get in touch with a representative today
Important Safety Information
THROMBATE III (antithrombin III [human]) is indicated in adult and pediatric patients with hereditary antithrombin deficiency for treatment and prevention of thromboembolism and for prevention of perioperative and peripartum thromboembolism.
Hypersensitivity reactions may occur. Should evidence of an acute hypersensitivity reaction be observed, promptly interrupt the infusion and begin appropriate treatment.
Because THROMBATE III is made from human blood, it may carry a risk of transmitting infectious agents, eg, viruses, the variant Creutzfeldt-Jakob disease (vCJD) agent, and, theoretically, the Creutzfeldt-Jakob disease (CJD) agent. There is also the possibility that unknown infectious agents may be present in the product.
Perform coagulation tests to avoid excessive or insufficient anticoagulation and monitor for bleeding or thrombosis. Measure functional plasma AT levels with amidolytic or clotting assays; do not use immunoassays.
In clinical studies, the most common adverse reactions (≥ 5% of patients) were dizziness, chest discomfort, nausea, dysgeusia, and pain (cramps).
The anticoagulant effect of heparin is enhanced by concurrent treatment with THROMBATE III in patients with hereditary AT deficiency. Thus, in order to avoid bleeding, the dosage of heparin (or low molecular weight heparin) may need to be reduced during treatment with THROMBATE III.
References
- THROMBATE III® (antithrombin III [human]) Prescribing Information. Grifols.
- AABB, American Red Cross, America’s Blood Centers, Armed Services BloodProgram. Circular of information for the use of human blood and blood components. June 2024. Accessed January 7, 2026. https://www.aabb.org/docs/default-source/default-document-library/resources/circular-of-information-watermark.pdf?sfvrsn=7f5d28ab_5.
- Rodgers GM, Mahajerin A. Antithrombin therapy: current state and future outlook. Clin Appl Thromb Hemost. 2023;29:10760296231205279.
- Tengborn L, Frohm B, Nilsson LE, Nilsson IM. Antithrombin III concentrate: its catabolism in health and in antithrombin III deficiency. Scand J Clin Lab Invest. 1981;41(5):469-477.125.
- Horstman E, Tormey C. Plasma products for transfusion: an overview. Ann Blood. 2022;7(4):1-13.
- Maclean PS, Tait RC. Hereditary and acquired antithrombin deficiency: epidemiology, pathogenesis, and treatment options. Drugs. 2007;67(10):1429-1440.
- Monagle P, Cuello CA, Augustine C, et al. American Society of Hematology 2018 Guidelines for management of venous thromboembolism: treatment of pediatric venous thromboembolism. Blood Adv. 2018;2(22):3292-3316.